News release
 Top
TopTOKYO, JAPAN - October 31, 2019 - Terumo Corporation (TSE: 4543) today announced that it has launched a joint research with Kikuna Memorial Hospital (Kanagawa, Japan) to develop artificial intelligence (AI) for Percutaneous Coronary Intervention (PCI). The company and hospital aim to develop an AI solution by around 2023, that will assist physicians to improve PCI accuracy and efficiency.
PCI is now a widely accepted less invasive procedure for treating ischemic heart diseases like angina and myocardial infarction, using various devices such as catheters and stents to open the clogged arteries. Physicians utilize angiographic and intravascular images to determine the most appropriate PCI strategy and procedure. However, a high level of skill is required to handle various medical devices, and to accurately make decisions for the best patient outcome.
In this collaborative investigation, both clinical images and patient data will be analyzed using AI. Terumo and the hospital will collaborate to develop technology that will assist physicians to improve PCI procedures. Specifically, the technology will assist physician's decision making by automatic measurement of the coronary artery diameter, plaque area, and optimal stent size.
Kikuna Memorial Hospital has years of experience and expertise in coronary intervention. The hospital will contribute their deep knowledge, and advice according to clinical practice.
Terumo offers a complete solution-based PCI product portfolio globally; from entry site management to lesion access of PCI. This new digital technology will add a new dimension to the portfolio, providing new value to interventional cardiologists. This study will be part of Terumo achieving their goal to cater an extensive solution to coronary intervention, and contribute to healthcare around the world.
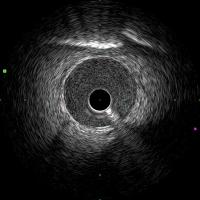
Intravascular ultrasound (IVUS) is used to guide stent implantation.
The figure shows typical IVUS image obtained using AltaView™ (Terumo Corporation).
About Kikuna Memorial Hospital
Kikuna Memorial Hospital is an acute care hospital located in Kanagawa, Japan. The hospital is accessible 24/7, taking in every emergency patient - summing up to over 7,000 ambulance-correspondence per year. The Cardiovascular Disease center, overviewed by director, Dr. Junko Honye, treats patients with a unique collaboration of interventional cardiologists and cardiac surgeons.
About Terumo
Terumo (TSE: 4543) is a global leader in medical technology and has been committed to “Contributing to Society through Healthcare” for 100 years. Based in Tokyo and operating globally, Terumo employs more than 30,000 associates worldwide to provide innovative medical solutions in more than 160 countries and regions. The company started as a Japanese thermometer manufacturer, and has been supporting healthcare ever since. Now, its extensive business portfolio ranges from vascular intervention and cardio-surgical solutions, blood transfusion and cell therapy technology, to medical products essential for daily clinical practice such as transfusion systems, diabetes care, and peritoneal dialysis treatments. Terumo will further strive to be of value to patients, medical professionals, and society at large.
