News release
 Top
TopTerumo Corporation (Headquarters: Shibuya-ku, Tokyo, Japan. President: Yutaro Shintaku) announced today that it has received conditional approval for manufacturing and selling its HeartSheet autologous skeletal myoblast sheets as a cellular or tissue-based products in Japan.
HeartSheet is cultured autologous skeletal myoblast sheets from a patient own muscle of thigh and transplanted to patients heart under the open chest surgery. This transplantation is expected to improve the patients heart condition significantly. A strong point of the therapy is the absence of any adverse reaction to the cells, since they are harvested from the patients own body.

Culturing cells at one of Terumo's facilities
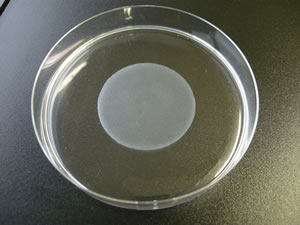
A skeletal myoblast sheet
The conditional approval requires:
I. 60 HeartSheet cases, showing efficacy of the product and treatment
II. Superiority compared to current existing treatment to 120 cases
III. Application for official approval within 5 years
Additional information:
- Terumo began developing cell sheets in 2007 as part of its R&D on cardiac regenerative therapy. It commenced a clinical trial at three medical institutions in Japan in 2012 and completed the study in 2014. On October 30, 2014, Terumo applied to Japans Ministry of Health, Labour and Welfare for approval to produce and market its skeletal myoblast sheets as a regenerative medicinal therapy for treating severe heart failure caused by chronic ischemic heart disease.
- Terumo has been promoting joint research with Osaka University, where clinical research on skeletal myoblast sheets is being carried out by Professor Yoshiki Sawa as part of a project sponsored by the New Energy and Industrial Technology Development Organization (NEDO). Terumo has also participated in a research project on regenerative medicine using cell sheets. It is led by Professor Teruo Okano of Tokyo Womens Medical University, which is part of a special consortia involved in developing advanced medical care in Japan.
About Terumo
Terumo (TSE: 4543) is a global leader in medical technology and has been committed to “Contributing to Society through Healthcare” for 100 years. Based in Tokyo and operating globally, Terumo employs more than 30,000 associates worldwide to provide innovative medical solutions in more than 160 countries and regions. The company started as a Japanese thermometer manufacturer, and has been supporting healthcare ever since. Now, its extensive business portfolio ranges from vascular intervention and cardio-surgical solutions, blood transfusion and cell therapy technology, to medical products essential for daily clinical practice such as transfusion systems, diabetes care, and peritoneal dialysis treatments. Terumo will further strive to be of value to patients, medical professionals, and society at large.
