News release
 Top
TopLAKEWOOD, Colo., U.S.A., and FORT COLLINS, Colo., U.S.A. - August 11, 2021 - Terumo Blood and Cell Technologies (Terumo), a medical device company specializing in a portfolio of technology, software and services for blood component collection, therapeutic apheresis and cellular technologies, and PhotonPharma Inc., a cancer immunotherapy developer, today announced they have established a memorandum of understanding (MOU) for collaboration to develop Innocell™, a novel tumor specific immunotherapy (a therapeutic vaccine) for solid tumors. The benefit to patients is that collaborations like this can help improve the speed to market for valuable treatments.
To help in the cancer immunotherapy development process, PhotonPharma is using Terumo's Mirasol® Pathogen Reduction Technology (PRT) in the manufacturing process. The initial agreement authorizes PhotonPharma to reference the Device Master File (DMF) on record with the FDA for the Mirasol system. This DMF will then support the PhotonPharma regulatory submission for its Innocell therapeutic vaccine technology. Additionally, Terumo Blood and Cell Technologies will supply Mirasol illuminators and single-use sets for the immunotherapy preparation process used in the clinical trial.
Mirasol has been used in select markets outside the United States since 2007. Terumo designed Mirasol to reduce pathogen load and inactivate residual white blood cells in whole blood and blood components. PhotonPharma's specific use of Mirasol is a further example of how Terumo's products are being used to develop treatments across the life-sciences sector to help patients gain access to medical therapies.
Another potential therapy in the War Against Cancer
The American Cancer Society's 2021 forecast projects 1.9 million new cancer diagnoses and more than 600,000 cancer deaths. Each day, that's approximately 5,200 newly diagnosed cancer cases and 1,670 cancer deaths. Ninety percent of the cases diagnosed are expected to be from solid tumors2.
Therapeutic cancer vaccines are developed to stimulate a patient's immune system to fight an established cancer. They are different from prophylactic vaccines, which are given to healthy individuals to prevent an infection and related disease.
PhotonPharma developed its technology to be differentiated from other therapeutic cancer vaccines in its potential to preserve solid tumor antigens, cell metabolism and protein translation while also potentially achieving inactivation of cellular replication.
This MOU is the first part of a longer-term collaboration to advance PhotonPharma's Innocell. The collaboration focuses on the development, regulatory approval and future commercialization of PhotonPharma's Innocell vaccine. PhotonPharma is currently preparing an Investigational New Drug (IND) submission related to a phase I clinical trial targeting triple-negative breast cancer.
"This agreement to work together with Terumo increases the potential to advance a new therapy for patients suffering from a variety of solid tumor malignancies including breast cancer," said Dr. Gary Gordon, M.D., Ph.D., former Divisional Vice President of Abbvie Oncology and PhotonPharma board member. "Working together will benefit patients in need of new therapeutic approaches to treat their underlying disease."
"For Terumo, strategic collaborations increase the potential speed to market and decrease development costs of therapies. This will enable patients to benefit from therapies earlier. Contributing toward the development and commercialization of Innocell enables Terumo to contribute toward the next potential major medical breakthrough." - Antoinette Gawin, President and Chief Executive Officer, Terumo Blood and Cell Technologies.
References
- 1
American Cancer Society. (2021). Retrieved 4 August 2021 from Cancer.org.
- 2
American Cancer Society. (2021). Cancer Facts & Figures. Retrieved from Cancer Facts Figures 2021: https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2021.html
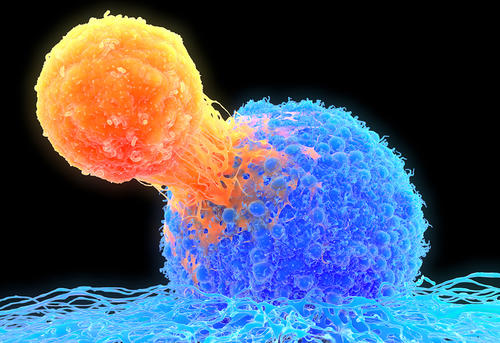
- Image Description
- T-cell attaching to cancer cell, illustration: T lymphocyte (orange) attached to a cancer cell (blue), illustration. T lymphocytes are a type of white blood cell that matures in the thymus. Certain kinds of T lymphocytes can recognize specific sites (antigens) on the surface of cancer cells or pathogens and bind to them. They can then destroy the cancer cells, or signal for other immune system cells to eliminate them. The genetic changes that cause a cell to become cancerous lead to the presentation of tumor antigens on the cell's surface.
About PhotonPharma
PhotonPharma is developing the Innocell therapeutic cancer vaccine, designed as a personalized therapy using autologous tumor tissue. The technology is based on a photochemical inactivation platform, designed to use riboflavin (Vitamin B2) and UV light in a proprietary method to render tumor cells incapable of replication but can still metabolize, express proteins, present antigen and stimulate immune recognition and response mechanisms.
In combination with selected adjuvants, PhotonPharma's Innocell has demonstrated the ability of this technology to prevent metastatic tumor formation, reduce the growth of tumors and stimulate cellular immune response pathways in preclinical development animal models.
PhotonPharma is based in Fort Collins, Colo., U.S.A., at the Research Innovation Center of Colorado State University. PhotonPharmaceuticals.com
About Terumo Blood and Cell Technologies
Terumo Blood and Cell Technologies is a medical technology company. Our products, software and services enable customers to collect and prepare blood and cells to help treat challenging diseases and conditions. Our employees around the world believe in the potential of blood and cells to do even more for patients than they do today.
Terumo Blood and Cell Technologies' customers include blood centers, hospitals, therapeutic apheresis clinics, cell collection and processing organizations, researchers and private medical practices. Our customers are based in over 130 countries across the globe. We have 750+ granted patents, with more than 150 additionally pending.
We have global headquarters in Lakewood, Colo., U.S.A., along with five regional headquarters, six manufacturing sites and six innovation and development centers across the globe. Terumo Blood and Cell Technologies is a subsidiary of Terumo Corporation (TSE: 4543), a global leader in medical technology. TERUMOBCT.COM
About Terumo
Terumo (TSE: 4543) is a global leader in medical technology and has been committed to “Contributing to Society through Healthcare” for 100 years. Based in Tokyo and operating globally, Terumo employs more than 30,000 associates worldwide to provide innovative medical solutions in more than 160 countries and regions. The company started as a Japanese thermometer manufacturer, and has been supporting healthcare ever since. Now, its extensive business portfolio ranges from vascular intervention and cardio-surgical solutions, blood transfusion and cell therapy technology, to medical products essential for daily clinical practice such as transfusion systems, diabetes care, and peritoneal dialysis treatments. Terumo will further strive to be of value to patients, medical professionals, and society at large.